Developmental Stages in Human Embryos
Go to Stage: Intro 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23
Stage 11
Page 105Approximately 2.5–4.5 mm
Approximately 24 ± 1 postovulatory days
Characteristic feature: 13–20 pairs of somites
SUMMARY
External: the rostral neuropore is in the process of closing; the otic invagination is shallow or still widely open; pharyngeal arches 1 and 2 are evident.
Internal: the sinus venosus develops; the oropharyngeal membrane ruptures; the mesonephric duct and nephric tubules appear; the optic vesicle and otic pit develop.
SIZE AND AGE
As for the size of the chorion, out of eight specimens five had largest diameters between 20 and 25 mm, two had largest diameters between 17 and 18 mm, and one was 15 mm. In average diameter (i.e., the mean of the largest and smallest diameters) six of these eight chorions fall between 15 and 18 mm. One is smaller than this (14 mm) and one is larger (21 mm). The size of the embryo itself is also variable, depending on the amount of shrinkage, manner of handling, and straightness or curvature of the specimen. Moreover, the accuracy of the measurement has to be taken into account. At this early stage it is difficult to use calipers without injuring the specimen. The most satisfactory measurements are those taken on reconstructions based on serial sections. Here, of course, one must allow approximately 25 percent for shrinkage. Omitting the obviously long and short specimens, the lengths of the sectioned embryos of this stage fall between 2.5 and 3 mm. An exceptionally long one measured 3.3 mm and an exceptionally short one measured 2.2 mm in length. When measured in formalin or 80 percent alcohol before sectioning, the embryos varied from 3.0 to 4.5 mm. One stretched in the linear axis was 5 mm long. Neither the relative size of the chorion nor the size of the embryo is constant enough to determine the level of development within the stage, although the embryo is the less variable of the two.
The age of embryos of this stage is believed to be approximately 24 ± 1 postovulatory days. The coital history in one instance (No. 1182b) gave an age of 26 days.
A detailed investigation of this stage, with particular reference to the nervous system, was published by Müller and O'Rahilly, 1986c, and similar studies of subsequent stages are in preparation.
EXTERNAL FORM
The criterion for stage 11 is the presence of 13–20 pairs of somites. This is the period of the delimitation and closure of the rostral neuropore. In the less advanced specimens of the group the fusion of the neural folds has extended rostrally to the region of the midbrain. In the more advanced specimens the neuropore is closing or has just completed its closure. An equally definite characteristic of the group is the presence of the mandibular and hyoid arches. The otic invagination can be recognized in most specimens as a slight depression, and in transparent specimens it can be clearly seen because of the refraction of its thick margins.
The general form of a less advanced member (13 somites) of the group is shown in figures 11-1 and 11- 2A, B. A more advanced member (19 somites) is shown in figure 11-2E in profile view, revealing its widely open communication with the umbilical vesicle. In the latter specimen there is a marked dorsal flexure, which is exaggerated here but commonly occurs in some degree in the more advanced embryos of this stage. As was pointed out by Bartelmez and Evans (1926), the more precocious head end and the heart tend to elevate that end. Similarly the relatively larger mass of the caudal end raises it above the level of the flattened, Page 106 thin umbilical region, and a moderate dorsal flexure should be regarded as normal (Orts-Llorca and Lopez Rodriguez, 1957). Later, as the spinal cord and body walls of this dorsal region take on more bulk, the flexure disappears, and a smooth, convex dorsal contour becomes permanently established.
The views shown in figure 11-2C, D illustrate details of the head and cardiac region, showing the appearance of the closure of the rostral neuropore and the form and relationships of the central nervous system. In formalin specimens the fifth and the acousticofacial nerves can usually be recognized. Although reconstructions have great value in reaching an understanding of the anatomy of young embryos, it is important also to form impressions of the character of the different tissues from study of the actual specimens and from good photographic reproductions, such as those shown in figure 11-2.
Drawings of eight representative specimens of this stage are shown in figure 11-3. These are enlarged to the same scale, so that the variations in size, posture, and proportions of the specimens can be observed. If it were not for the unavoidable handling of the specimens, it appears likely that there would be but slight variations in these respects. The neural tube is the chief determiner of the form of the embryo at this time. For that reason it is shown in median section, whereby its parts and topography are made more readily recognizable. It will be seen that the dorsal flexure occurs at the transition from the larger and more precocious rostral end of the neural tube to the smaller, newly closed tube that terminates in the caudal neuropore. This flexure can be seen in some degree in all specimens of 17–20 pairs of somites.
The surface ectoderm at stage 11 possesses areas of at least three different thicknesses, and the ectodermal ring begins to form as an inverted U (O'Rahilly and Müller, 1985).
HISTOLOGICAL FEATURES
Primitive streak. The caudal eminence, which lies between the cloacal membrane and the site of the neurenteric canal, represents the region of the former primitive streak. It contains dense axial material from which new parts of the notochord are added, and paraxial condensations from which the somites develop (Müller and O'Rahilly, 1986c).
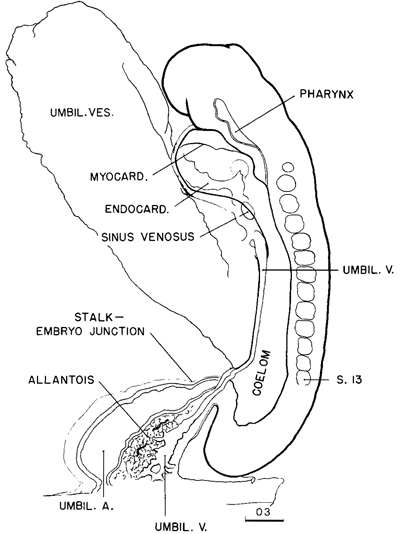
Fig. 11-1. Outline drawing of the left lateral view of No. 6344 (13 pairs of somites), showing the features that can be identified in the photograph of the right lateral view (fig. 11-2A).
Somites. Four pairs of somites are considered to be occipital, although it is only in the next stage that the neural crest for the cervical region is clearly delineated and can be used as a criterion for distinguishing the occipital from the cervical region.
Somite 1 is small and makes almost no contact with the surface ectoderm. It is situated immediately caudal to the vagal-accessory neural crest. Numerous mitotic figures in its walls indicate the beginning of its transformation into material for the hypoglossal cord (O’Rahilly and Müller, 1984b). In general, the rostrocaudal extent of an individual somite is variable. Calculations show that a clear gradient in size between rostral and caudal regions is lacking. In most cases the rostral somites are larger in embryos with 13 as well as in those with 20 somites. The mean time for the formation of one pair of somites during stages 9–12 inclusive is 6.6 hours (Müller and O'Rahilly, 1986c).
Page 107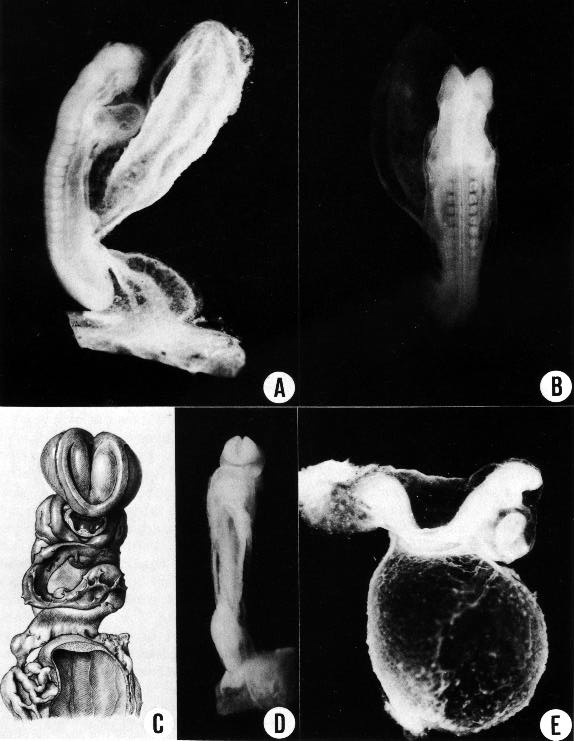
Fig. 11-2. (A) Right lateral view of No. 6344 (13 pairs of somites).
(B) Dorsal view of the same embryo, showing that neural closure has advanced rostrally to the area of the trigeminal nerve. The somites and spinal cord are distinguishable. The median dark line that extends along the spinal cord is the transparent central canal. On each side of it is a narrow, white, opaque strip, which is the line of greatest thickness of neural tissue. Latter to it the cord rounds off and to that extent is thinner.
(C) No. 4529 (14 pairs of somites), based on a reconstruction by Osborne O. Heard. It shows how the neural lips begin to dose rostrally and come to cover D2.
(D) No. 6784 (17 pairs of somites), showing a later phase in the closure of the rostral neuropore, with the typical blunt end of the neural tube.
(E) No. 6050 (19/21 pairs of somites), showing the umbilical vesicle. A dorsal curvature is common at this time. It is caused by the flexibility of the thin central part of the embryo as contrasted with the two ends, the bulbous caudal tip with connecting stalk at the rear, and the advanced head structures and heart rostrally. This specimen shows the entrance through which the coelomic fluid gains access to the mesoblast. By chance there was a break in the umbilical vein and some embryonic blood escaped into the cardiac region of the coelom, delineating its dorsal contours, as though injected. As is typical of this stage, the umbilical vesicle still opens widely into the gut.
Page 108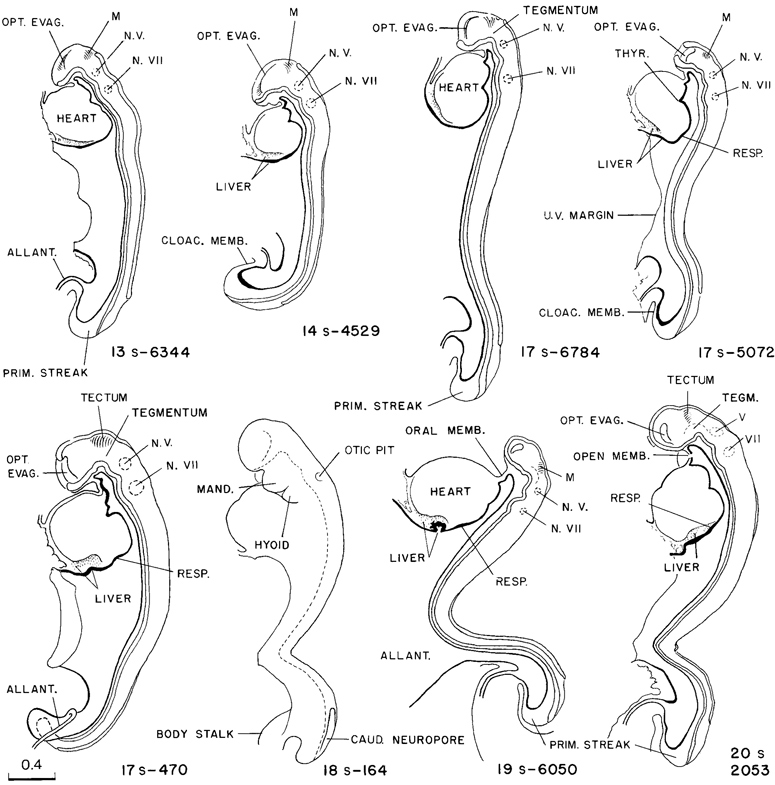
Fig. 11-3. Eight representative specimens of stage 11, enlarged to the same scale and hence directly comparable as to size, proportions, and posture. It will be noted that the neural tube appears to be the chief factor that determines form, and the primary neural parts are already distinct. The number of somitic pairs and the collection number of the embryo are given for each specimen. The tracing of the 18-somite embryo. No. 164, was made from a photograph. All the others are median section drawings made from Born reconstructions. It will be seen that active proliferation in the hepatic region is beginning in the more advanced members of the group.
Page 109Coelom. Streeter, whose detailed description of the coelom follows, defined the term coelom as “any walled off space or fluid reservoir uniformly present in the mesoblast” and “also any similar space formed in the extra-embryonic mesoblastic reticulum.” Furthermore, under the term coelom “are to be included both the free passage for coelomic fluid and the enclosing walls that are irrigated by it.” This idea has been explored further in the rat by Langemeijer (1976), who writes of a “coelomic organ,” i.e., a single, tubular organ serving as the blastema of various thoracic organs and comparable to the neural tube in possessing a specific wall that encloses a cavity.
When mesoblastic cells become detached from the pluripotential cells of the primitive streak, they are advanced one degree in specialization, which means that they are therewith restricted to certain potentialities in respect to development; they no longer share with the epiblast the possibility of becoming skin ectoderm or neural ectoderm. They can still take on many forms, however. As they proliferate and spread out in the form of a reticular sheet, some of them can soon be recognized as vasoformative strands, representing the elements of the blood vascular system.
The first conspicuous event in mesoblastic organization, however, is the beginning bilateral segmentation of the medial part of the mesoblast along its longitudinal axis. On each side, along the neural tube, focal centers of active differentiation occur serially, and there are thereby produced the highly characteristic somites. In its more peripheral regions the mesoblastic sheet shows no trace of segmental development. Instead, the proliferation of mesoblastic cells produces a thickened marginal band in which the cells become arranged as coalescing vesicles, resulting in a continuous mesoblastic cavity or passage. This development first occurs in a crescentic field at the rostral end of the embryonic disc. Such a passage is already present in stage 9 and, in stage 10, it has extended caudad and opens freely on both sides into the extra-embryonic coelom. In this manner an advantageous arrangement is provided by which the more deeply lying mesoblastic parts of the embryo can be freely reached by the coelomic fluid, which at this time is the sole source of nutriment. Although this is but a primitive type of circulation, it appears to serve that function adequately up to the time it is superseded by the blood vascular system. In other words, in the early organization of the mesoblastic tissue the first two major events are the differentiation of the medial part as segmental somites and the differentiation of the peripheral part as the non-segmental coelom. The coelomic walls respond with characteristic activity, and later give origin to various kinds of structures.
By stage 11 the coelom has become a conspicuous feature. Its distribution in a 13-somite embryo is shown in figure 11-4. In this drawing, slabs have been removed, exposing schematically three representative levels: upper cervical level, somite 4, and somite 8. It will be seen that below the level of somite 6 there is a free opening from the exocoelom into the intra-embryonic coelom in the cleft separating the umbilical vein and the primordium of the abdominal wall from the umbilical vesicle. Caudally, the coelom extends along the side of the intestinal epithelium and spreads caudad with it as the caudal bud continues its differentiation, always remaining widely open to the exocoelom and communicating with the opposite side across and caudal to the junction of the gut and umbilical vesicle. Above, at the level of somites 5 and 6, a trace of the multiple openings from the intra-embryonic coelom into the exocoelom, which originally characterized this system, still persists in the form of a partition marking off an upper opening from the main communication. In the more advanced specimens of this stage, this partition disappears, and there remains but one common opening. Following the coelom rostrally, it is seen again as a closed passage at the level of somite 4, and rostrally from there it soon widens out into the original coelomic space, which now invests the endothelial heart and its gelatinous envelope. It would seem that the first cardiac movements would tend to facilitate the flow of fluid through the coelomic passage.
Typical transverse sections through the same 13-somite embryo are shown in figure 11-5. It will be seen that everywhere along the margins of the coelom an active proliferation of cells is taking place, more active in some regions than in others. In addition to proliferating, these mesoblastic cells also are differentiating into special tissues that will constitute the connective Page 110 tissues, muscle, and vasoformative elements of the heart, lungs, and alimentary canal. Thus in section A, through the rostral end of the heart, the visceral part of the coelomic wall is producing myocardium and the parietal part is shedding cells that will form various elements of the thoracic wall. The definitive pericardium will be formed by the residual coelomic cells after the more special cells are segregated out. As for the pleurae, they are even more remote. Photographs showing early histogenesis of the myocardium are reproduced as figure 11-6A, D. It will be seen that the visceral surface of the coelomic passage does not exist as a membrane, separate from the myocardium; instead, the surface cells merge directly into the more deeply lying cells. The surface cells are more-actively proliferating and remain more primitive than the deeper ones, whereas the deeper ones are on the way to specialization and to that extent have sacrificed their potentialities. The surface cells do not yet exist as a distinct and separate layer.
Proceeding to the level of somite 1, section B of figure 11-5 passes through the base of the heart and obliquely through the primordium of the liver. In respect to the heart, the section shows the sinus venosus and the area where the heart rests on the liver. It will be noted that the coelom is actively giving off cells that form a condensed mass constituting the early mesoblastic framework of the liver. Photographs illustrating this process under higher power in a slightly more advanced embryo are shown as figure 11-6A–C. Note is to be taken of the transition of the coelomic surface cells into the deeper cells and the phenomenon of angiogenesis that immediately begins in the latter (fig. 11-6B). The digestive epithelium lies under the more dorsal part of the hepatic primordium, and doubtless is playing a role in its development, but it is not until the next stage that a really distinct hepatic diverticulum and the origin of hepatic epithelial cords that penetrate the mesodermal framework provided by the coelomic wall become apparent. Figure 11-6B shows the digestive epithelium underlying the hepatic primordium shortly before the epithelial cords invade the liver. Thus, in the formation of the liver its mesodermal elements are supplied by die coelom, whereas its epithelial elements come from the opposite direction (i.e., from the gut).
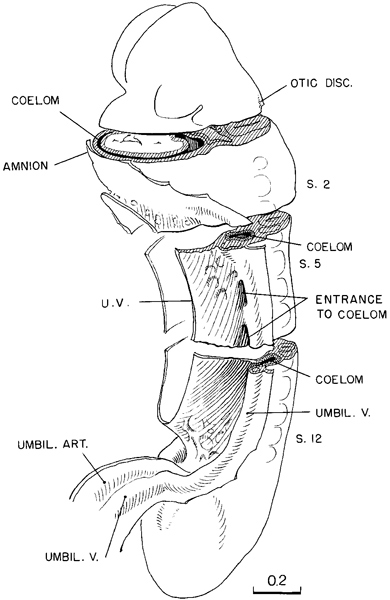
Fig. 11-4. The distribution of the coelom in a 13-somite embryo (No. 6344). At three representative levels die surfaces are exposed schematically, showing the relations of the coelomic channel to the mesoblast and to the heart. The first pulsations of the latter presumably aid in the movement of coelomic fluid, which at this time is the sole source of nutriment for these tissues.
At the level of somite 4 (fig. 11-5C), not only does the coelom provide a passage for its contained fluid, but also its walls are actively proliferating cells that are to take a large part in the formation of the gut wall, the adjacent vessels, and the body wall itself. Further caudally (section D) is shown the free communication of the intra-embryonic coelom with the exocoelom. It will be seen that coelomic proliferation is in general the same at all levels. The proliferation and accompanying specialization of these cells are roughly comparable to what is seen in the differentiation of the neural tube. There too the germinal cells lie near the central canal, and the segregated special cells (mantle zone), produced in the course of proliferation, move away from the border of the lumen. It is also to be noted that certain fields of this coelomic proliferation are sharply marked off from one another.
Page 111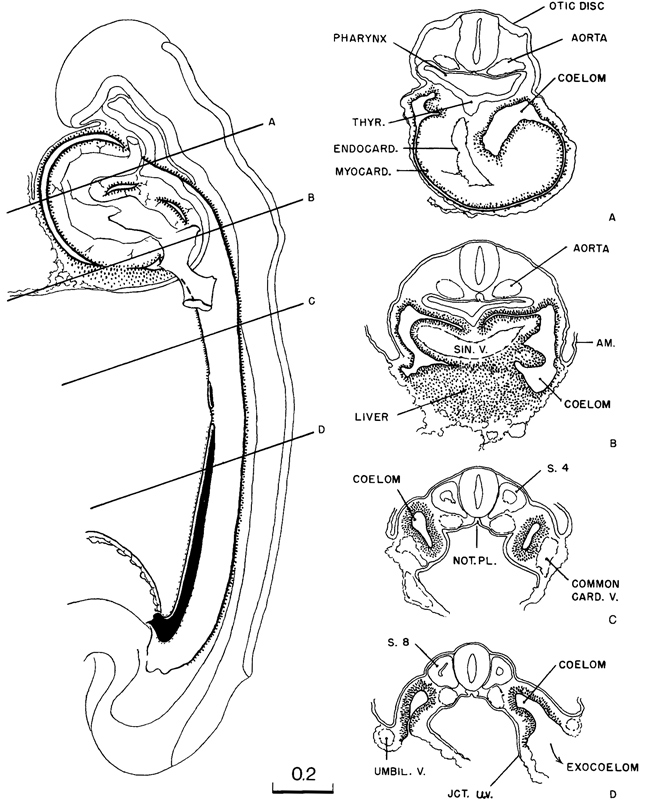
Fig. 11-5. Typical sections through the coelom of No. 6344. The mesoblastic surface bathed by the coelomic fluid is a locus of active cell proliferation. It is a germinal bed from which the specializing daughter cells move inward radially to form various structures according to the field from which they arise. Thus the coelomic walls give origin to such divergent tissues as myocardium, the framework of the liver, and the muscular coats, blood vessels, and connective tissue envelopes of the gut. The coelomic walls are demarcated at this stage into territories each of which has its own characteristics.
Page 112
Fig. 11-6. (A) Section showing the hepatic and cardiac regions in a 16-somite embryo (No. 7611, section 3-4-1).
(B) Same section, area enclosed by white line in A. This is the hepatic primordium. The cells that are to form the framework are proliferating at the coelomic surface and crowding downward. Some of them are already undergoing the first steps in angiogenesis. Below is the gut epithelium, which is not yet giving off epithelial cords.
(C) Section at a more rostral level (3-2-6) of the same specimen. Framework cells are proliferating from the coelomic surface and show early angioblasts. This is just in front of the junction between the gut and umbilical vesicle, and no gut epithelium is included.
(D) Detail of the myocardium in the same embryo (section 3-3-2). The coelomic fluid (asterisk) comes in contact with the hepatic region below, and it bathes the surface of the folded myocardium above. The surface cells of the myocardium constitute a germinal bed from which specialized myocardial cells are arising. Differentiation follows rapidly. Aside from the germinal bed, there is no surface membrane that can be designated as pericardium, which will appear later.
Page 113After this orientation on the structure and topography of the coelom, it is easier to understand the surface anatomy of embryos of this stage as seen in photographs. Specimens at this time are quite transparent after formalin fixation, and by means of stereoscopic photographs much of the anatomy can be observed. A large part of the coelomic passage can be seen in the specimen shown in figure 11-2A. This is to be compared with the accompanying explanatory diagram (fig. 11-1). A more advanced specimen (19 somites) is illustrated in figure 11-2E. On each side of this embryo, a single and wider opening from the exocoelom into the intra-embryonic coelom can be plainly seen. By a fortunate accident, there was a slight extravasation of embryonic blood, apparently from a tear through the umbilical vein, and this blood found its way into the cardiac region of the coelomic passage in such a way as to outline the dorsal confines of the cavity as though it were injected. Such an opening into the coelomic passage provides ready access of the exocoelomic fluid to the deeper tissues of the embryo. In the next stage the opening is still larger.
The coelomic epithelium is exceptional during early development in its close structural and histogenetic relationships to the underlying mesenchyme. The capacity of embryonic coelomic cells to change from epithelial to mesenchymal, or vice versa, can be seen in the development of their derivatives. Thus, in the initial formation of the gonads (and the suprarenal cortex), the coelomic wall reverts to an early condition in which the surface lining acts as a germinal layer and produces mesenchymal cells, the basement membrane of the mesothelium being abolished (Gruenwald, 1942). The blastema thereby formed differentiates into the epithelial primary sex cords and the intervening mesenchyme (ibid.).
Notochordal plate and notochord. The development of the notochordal plate into notochord proceeds longitudinally from caudal to rostral, and the last areas to retain the plate are in the pharynx. The separation of the notochordal plate from the alimentary system involves the following: (1) the plate becomes U-shaped even in stage 10, (2) the vertical limbs of the U begin to move medially and touch each other, (3) the adjoining digestive epithelium begins to form a thin cytoplasmic bridge, and (4) mitotic figures appear and the still U-shaped notochord is cut off entirely (Müller and O'Rahilly, 1986c).
The part of the notochord that forms directly from the axial condensation in the caudal eminence is far thicker and more advanced in differentiation. The notochord has a dual mode of origin: (1) rostral to the neurenteric canal or its site, it develops from the notochordal plate, and (2) caudally it arises from the axial condensation.
The notochordal plate and notochord are still intimately related to the neural tube (and the notochord to the digestive epithelium), and their basement membranes are in contact.
Neurenteric canal. The former site of the canal can generally be determined but with much more difficulty than at the previous stage. The site moves caudad during stages 10 and 11.
Prechordal plate. The prechordal plate (Gilbert, 1957), which is more difficult to recognize than in previous stages, lies adjacent and rostral to the notochord, and caudal and lateral to the adenohypophysial area. Scant prechordal material is found in the median plane, most having migrated laterally to form the premandibular condensation.
CARDIOVASCULAR SYSTEM
Not only is the heart beating but “it is generally accepted that peristaltic flow begins” during stage 11 (de Vries and Saunders, 1962).
A description of the heart and blood vessels present in a 14-somite embryo has been given by Heuser (1930) and in a 20-somite specimen by Davis (1923). Here attention will be confined to the general characteristics of a vascular system that is adequate for an organism having the size and complexity found at this stage.
In figure 11-7 are shown in outline the endothelial vessels and plexuses that characterize the embryo at the time when its heart has begun to beat. The vascular apparatus still consists of simple endothelium. Only in the cardiac region have auxiliary tissues been differentiated. The endothelium of the heart is enclosed in a jelly-like envelope, as has been well described by Page 114 Davis. This cardiac jelly is in turn enclosed by a mantle of contractile tissue, the primordium of the myocardium. The character of this myocardium is shown in figure 11-6A, D. It is to be remembered that the myocardium is a specialization of the coelom, and its outer surface is bathed directly by the coelomic fluid. The immediate consequence of its specialization is its beginning pulsations.
Returning to figure 11-7, it will be seen that the vascular system of the caudal half of the embryo consists of a changing capillary plexus, which along with the laying down of the caudal end of the embryo spreads backward, always readjusting its communications with the large vascular channels of the connecting stalk. In the rostral and more advanced half of the embryo, the endothelial channels are attaining a more mature pattern. As would be expected, the endocardium and its associated coelomic derivatives, which originally were situated at the rostral end of the embryonic disc, show the most-advanced differentiation. The embryo is thereby provided at this time with a mechanism that serves to stir, and thereby aid the diffusion of, the substances contained both in the fluid of its endothelial plexus and in the coelomic fluid. Although it is too early to speak of a true circulation, under the conditions of an ebb and flow, there is a directional tendency from the heart to the aortae and from the umbilical vesicle to the heart. The capillaries of the rostral part of the umbilical vesicle, where it joins the gut endoderm, have already responded to the precocious endocardium by active growth and adjustment in the form of enlarged channels, which communicate with the heart. In a 16-somite embryo a sinus venosus is already present, and in more-advanced specimens of this stage a constriction begins to mark the boundary that separates it from the atrium proper. The cardiac pulsations may well be a factor in this endothelial adjustment. Although the plexus of the umbilical vesicle is not well advanced at this stage, one can distinguish at its rostral end the numerous communications that will later result in the omphalomesenteric veins, and likewise the mesh from the aorta that will produce the omphalomesenteric arteries. The umbilical veins and arteries are already formed; for this development, the connecting stalk and allantoic influences appear to have been responsible. The primary blood vessels of the central nervous system are more or less connected up in the more advanced parts, but the cardinal veins and the segmental branches from the aorta and their spinal cord communications are incompletely connected. At the caudal end of the embryo, they are represented merely by isolated vesicles. The common cardinal veins do not yet bridge the interval to the atrial endocardium, and a true circulation must await that development.
The fluid throughout the endothelial system, during this ebb-and-flow period, contains relatively few cells. A survey was made by Streeter of serial sections of thirteen embryos of this stage. For the most part the vessels were free of cells in the rostral half of the embryo, including the atria, ventricles, aortae, and umbilical veins. Occasionally, a few scattered embryonic blood cells or small clumps of them could be seen. In the caudal half, the aortae and the umbilical arteries and veins of the connecting stalk either were empty or contained a few cells. In some, numerous cells or clumps of cells were noted. In two specimens both the caudal region of the aorta and the umbilical arteries were moderately distended with blood cells, as though the cells had backed in from the umbilical vesicle. In one specimen the umbilical vein on one side was moderately distended with blood cells. This does not signify that the blood had escaped from the embryo into the chorion, because the latter likewise was largely free of blood cells. The villi contained very few cells. The vessels of the chorionic membrane, usually the branches of the umbilical artery, contained a moderate number of cells, but none of them was distended. The umbilical vesicle showed uniformly the presence of active blood islands, close together in the collapsed sac and separated by intervals of tenuous wall when the vesicle was distended. In most regions of the umbilical vesicle, all capillaries were filled with blood cells. In the proximal part of the sac, where it is continuous with gut endoderm, one frequently finds large capillaries completely empty or nearly so. It is such connecting branches that open into the sinus venosus and the cardiac end of the umbilical vein.
In addition to the rather complicated vascular system described above, one finds a differentiation of many new foci of angiogenesis, particularly along the surface of the central nervous system and in other mesoblastic regions In figure 11-6B is shown an early stage of angiogenesis in the coelomic mesoblast of the hepatic Page 115 primordium. The segregation of specialization of these angioblastic cells apparently occurs during proliferation of the coelomic cells. Among these new cells are some that become blood-forming cells, and these become set apart from the less specialized embryonic connective tissue cells, all of which, however, are destined to form the final framework of the liver.
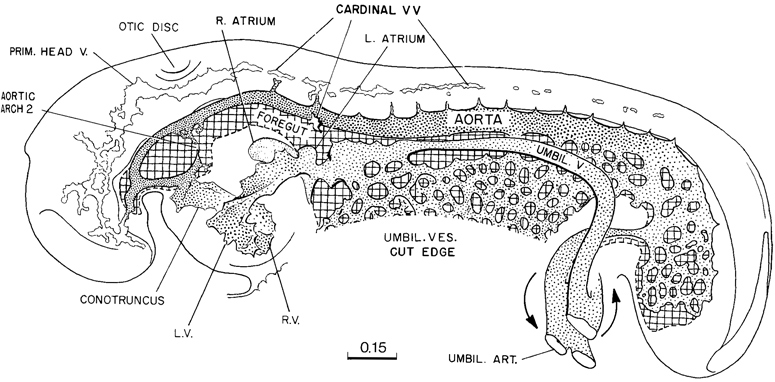
Fig. 11-7. Simplified drawing of the vascular system of a 14-somite embryo (No. 4529). The heart was probably pulsating when the embryo was removed. The primitive plexiform pattern would indicate an ebb-and-flow type of circulation. At the cardiac end of the plexus of the umbilical vesicle, the channels are beginning to be specialized into trunks characteristic of the venous end of the heart. The central nervous system, because of the widely open neuropores, is not yet dependent on a blood supply, and one can see only the first elements of a cardinal system. This drawing is based on the reconstruction published by Heuser (1930).
In short, one can say that at this stage the embryo is adequately maintained by a simple system of large endothelial channels that course through the deep tissues, and that in these channels an almost cell-free fluid ebbs and flows because of the action of the pulsations of the myocardium. What is necessary in the way of regulation of osmotic pressure and nutrient supply is provided in this manner, over and above the diffusion already secured by the services of the coelomic fluid that irrigates the various areas of the coelom.
By the end of stage 11 (fig. 15-5) the sequence of the cardiac chambers is: sinus venosus, right atrium, left atrium, atrioventricular canal, left ventricle, right ventricle, conotruncus, aortic arches. The “sinus venosus has almost completed its separation from the left side of the heart and opens into the dorsum of the right atrium, a fundamental relation which it ever after retains” (Davis, 1927). The ventricles have descended relative to the atria (Müller and O'Rahilly, 1986b), and the atrioventricular canal has changed from a vertical to a dorsoventral orientation (Davis, 1927). The ventricles have become trabeculated and the interventricular septum may have appeared (McBride, Moore, and Hutchins, 1981). The conus cordis has differentiated from the right ventricle (de Vries and Saunders, 1962), and the aortic sac (or sinus) develops as additional aortic arches form (deVries, 1981). The dorsal mesocardium has ruptured.
DIGESTIVE AND RESPIRATORY SYSTEMS
Pharyngeal arch 2 develops and arch 3 may do so. Pharyngeal membrane 2 appears and pharyngeal pouch Page 116 4 may form. The thyroid primordium has been subject to varying interpretations (O'Rahilly, 1983a). The oropharyngeal membrane may have begun to rupture. Esophagorespiratory and hepatorespiratory grooves may be found, the hepatic diverticulum grows into the septum transversum, and the cystic primordium may become distinguishable.
The pulmonary primordium is more evident than previously and it shows a high alkaline phosphatase reaction (Mori, 1959b).
URINARY AND REPRODUCTIVE SYSTEMS
The mesonephric duct develops as a solid rod in situ from the nephrogenic cord (Torrey, 1954) or perhaps from ectodermal buds lateral to somites 8–13 (Jirásek, 1971). The nephrogenic tissue develops into nephric vesicles which are connected by (at first solid) tubules with the mesonephric duct.
The cloacal membrane is in a central, oval depression on the ventral surface of the caudal part of the body wall.
The primordial germ cells are migrating from the umbilical vesicle to the hindgut (Witschi, 1948).
NERVOUS SYSTEM
The brain at stage 11 has been described in detail by Müller and O'Rahilly (1986c).
The neural tube appears to be the chief factor that determines the form of the embryo at this time. All embryonic organs seem to be influenced in their form by their environment, but of them the central nervous system is perhaps the most nearly independent. Its form seems to be the unmixed expression of the proliferation and enlargement of the cells that compose its wall.
The closure of the rostral neuropore in stage 11 (figs. 11 -3 and 11-8) is basically a bidirectional process: it continues from the rhombencephalon to the mesencephalon and proceeds from the optic chiasma toward the roof of D1, thereby forming the “adult” lamina terminalis and the future commissural plate. In embryos with 13 somites the rostrocaudal closure has scarcely begun, and hence the material of the future lamina terminalis still lies laterally. By 14 somites, closure at the level of the lamina terminalis has commenced. In embryos having about 20 pairs of somites, the forebrain is completely closed.
When the neural ectoderm becomes shut off from contact with the amniotic fluid, compensation is simultaneously made by the development and spread of blood capillaries, and these promptly invest the neural tube with a close-meshed network. Later, as the neural wall becomes thicker, this capillary network responds by sending in branches, thereby bringing the required service to the more deeply lying cells. The skin ectoderm continues to obtain what it needs from the amniotic fluid and for a long time requires no special provision in the way of capillaries.
A typical example of brain form in embryos of this stage is shown in figure 11-9. The drawing of a well-preserved 16-somite embryo should be compared with the dorsal view shown in figure 11-2B (See also fig. 13- 10.) The forebrain, consisting of D1 and D2 (already present in stage 10, see fig. 10-7), is still relatively simple. Part D1 is largely the area of the optic primordium and, in the median plane, the chiasmatic plate. The caudal limit of the plate is sometimes indicated by a postoptic recess. The floor of D2 includes mamillary and neurohypophysial components, and the latter is recognizable indirectly by the primordium of the adenohypophysis, which is the ectodermal region at the summit of the oropharyngeal membrane. Part D2 still protrudes toward the median plane, and its right and left convexities almost touch each other in the least advanced embryos. As the optic primordia become more evaginated, the walls of D2 recede and the ventricular cavity begins to expand. The medial parts of the forebrain rostral to D1, which fuse during the closure of the rostral neuropore in stage 11, are telencephalic. They contribute to the formation of the embryonic lamina terminalis and commissural plate.
The most uncertain part to identify precisely is the mesencephalon (tectum and tegmentum in fig. 11-9) because its delineation toward both the diencephalon and the rhombencephalon is difficult. It appears to consist of only one segment at this stage.
Neuromeres, which are transverse swellings in the developing neural tube, can best be assessed by evaluating the three main planes of section. Swellings and grooves are best shown in reconstructions of sagittal sections, whereas transverse sections are preferable in Page 117 determining the relationship to ganglia. Indications of neuromeres are already evident at stages 9 and 10. A combination of the various data shows two neuromeres in the forebrain (D1 and D2), one in the midbrain, and 6-7 in RhA–RhC. Part RhD, the area of somites 1–4 (the hypoglossal region), is clearly delineated neither by a groove nor by a deepening. D1 and D2 correspond to neuromeres a and b, M to proneuromere B, RhA, RhB, and RhC to proneuromeres C, D, and E of Bergquist and Kallen (personal communication, 1969). Their inventory contains no equivalent for RhD of the present authors. RhA–RhC, as described by Bartelmez (1923) for stage 9, were termed proneuromeres by Bergquist (1952) because of their existence in the still open neural groove. Three-dimensional reconstructions demonstrate the neuromeres clearly to be serial swellings of the brain.
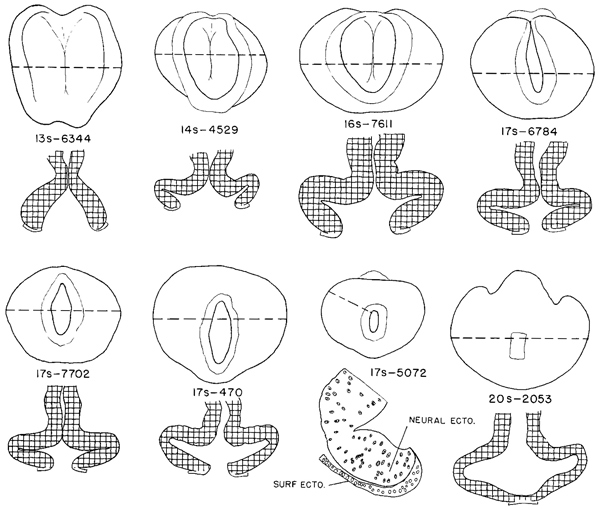
Fig. 11-8. Outline drawings of the rostral end of the neural tube in a series of embryos, showing the steps in the closure of the rostral neuropore. Underneath each specimen is shown a horizontal section taken at the level indicated by the broken line. These show the extent of the optic evaginations. In the section of the 17-somite specimen (No. 5072) only the right side is drawn, showing the demarcation between neural and skin ectoderm. With the exception of No. 5072, all are drawn to the same scale.
The rhombomeres can be distinguished comparatively easily by the developing ganglia (fig. 11-9). The trigeminal ganglion characterizes Rh2, the faciovestibulocochlear ganglion Rh4, the glossopharyngeal Rh6, and the vagal Rh7; Rh3 and Rh5 appear to have no neural crest. Somites 1–4 enable RhD, the region of the hypoglossal nerve, to be identified.
The picture presented by the walls of the spinal cord at this time reveals, by the frequency of mitotic figures, that the germinal bed of proliferating cells lies along the margin of the central canal. It is from the canal that nutriment still diffuses among the cells. From this germinal bed new cells move externally. Cell proliferation and the concomitant cell specialization is only in its early phases at this stage, and one cannot yet mark off ependymal and mantle zones, such as characterize later stages.
Page 118Neural Crest
Neural crest cells are still being given off at the site of Rh4, Rh6, and Rh7, all of which are now closed. Therefore, the same areas, open in stage 10 and producing neural crest already at this former stage, continue the production of neural crest material when closed. What is the same in both modes is the fact that the neural crest cells are clearly derived from the neural ectoderm.
Neural crest cells contribute to the formation of head mesenchyme together with the material from the prechordal plate. The mesencephalic neural crest is still being produced and spreads toward the optic evagination, and, in more-advanced embryos, mixes with optic crest cells thereby forming the sheath of the optic vesicle. The trigeminal neural crest is more condensed than in stage 10. A condensation of cells is visible within the mandibular arch, but it is not possible to decide whether it is neural crest material deposited there during stage 10, or paraxial mesenchyme, or both. Also in pharyngeal arch 2, two different groups of cells seem to be present, the first clearly representing ganglion 7/8, and the second, also slightly more condensed, forming a prolongation within the arch. Prechordal mesenchyme was spreading out laterally in the premandibular area and ventrally to the heart in stage 10; it is now limited to the premandibular area.
The nasal plate is one of the ectodermal areas giving rise to neural crest. Although it does so only in later stages, the plate becomes apparent in stage 11 and lies on both sides of the neuropore (O'Rahilly and Müller, 1985).
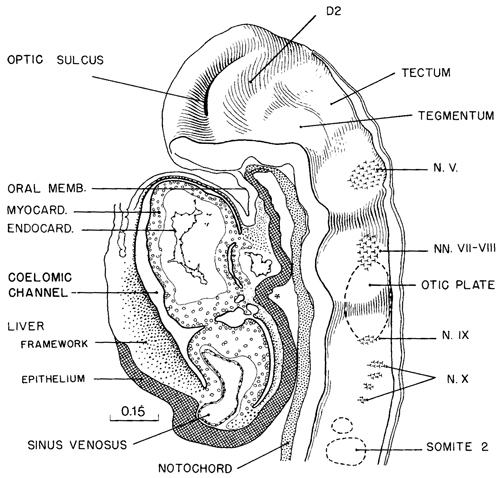
Fig. 11-9. Median view of the rostral half of a 16-somite embryo (No.7611), showing segmental structures in the forebrain (D1 and D2) and in the hindbrain The area of ganglion 5 is rhombomere A, the area of ganglion 7/8 is RhB, and the area of ganglia 9 and 10 is RhC. The adjacent structures that participate in giving form to this part of the embryo are shown in the drawing. The median ventral pocket of pharyngeal epithelium, marked with an asterisk, indicates the beginning of the thyroidgland.
The histological character of the neural tube in a 17-somite embryo is shown in figure 11-10. The sections A and B are taken at two different regions. The upper is at the level of somite 5, cutting obliquely through the spinal cord and thereby exaggerating its ventrodorsal diameter. Compared with the section through the lower level (somite 16), it is larger; its walls are thicker and contain more cells, and the central canal is correspondingly compressed. Another Page 119 feature showing advance in maturity is the migration of the neural crest cells, which have detached themselves from the cells of the dorsal lips at the closure line. At the more caudal level the neural crest cells have not started their migration, and because of their presence the dorsal seam has the gross appearance of a keystone closing in the arched lateral walls. These crest cells do not appear to be proliferating as actively as the other cells of the neural wall, and the wedge-shaped area formed by them is correspondingly paler. It is after their migration that the active proliferation of the crest cells occurs. The progress of neural crest formation has been plotted for stage 11 by Bartelmez and Evans (1926) and by Heuser (1930).
Eye
The right and left optic primordia meet at the optic chiasma and, together with the latter, form a U-shaped rim in the least developed embryos of stage 11 (fig. 11-2C).
The optic evagination is produced at the optic sulcus when about 14 pairs of somites are present, and the optic ventricle is continuous with that of the forebrain (O'Rahilly, 1966, 1983b). The optic evagination constitutes the optic vesicle when approximately 17–19 pairs of somites are visible (fig. 11-8).
The wall of the optic evagination contributes neural crest to its mesenchymal sheath from about 14–16 pairs of somites onward (Bartelmez and Blount, 1954). The sheath probably also acquires cells from the mesencephalic neural crest (Müller and O'Rahilly, 1986c). The sheath then separates the evagination from the overlying ectoderm. A caudal, limiting sulcus develops between the optic evagination and the forebrain.
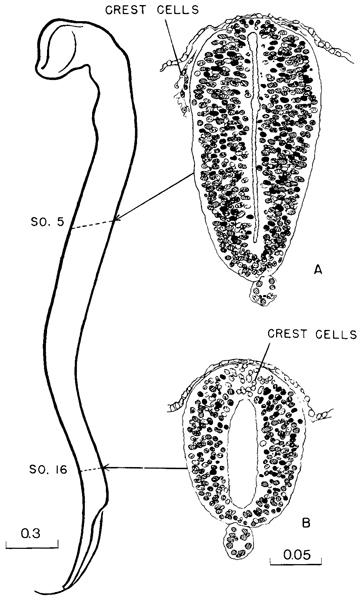
Fig. 11-10. Contour of the neural tube of a 17-sornite embryo (No. 6784). The upper two-thirds is definitely more advanced than the recently closed lower third. Thus section A, through the level of somite 5, is more mature than section B, through somite 16. In the former the neural crest cells have migrated ventrally, whereas at the more caudal level they are still incorporated in the neural wall.
Ear
The invaginating otic disc is already a sharply demarcated structure, and with suitable illumination it can be seen in a gross specimen. It is seen to best advantage, however, in stained sections. A series of such sections is shown in figure 11-11, and they cover the range of development that characterizes this stage. The definite line that marks the junction of the disc with the skin ectoderm is characteristic of this stage. It is to be noted that this feature is acquired before adistinct pit appears. The disc may be regarded as an island of neural tissue that has the appearance of having floated away from the main neuroectodermal mass and is now separated from it by the intervening skin ectoderm. During its proliferative period, this disc of specialized neural ectoderm closely resembles the parent neural tube. To go further back, it is to be remembered Page 120 that the neural ectoderm is a product of the proliferation of the pluripotential mother cells of the epiblast. The neural ectoderm becomes segregated by stage 8 and becomes one of the primary embryonic tissues. As it does so, the central strip laid down becomes the neural tube with its particular potentialities. It is possible that at the same time some of the marginal neuroectodermal cells are specialized in the direction of forming various plates and neural crest masses. These become detached, some earlier and some later, from the main neural plate, and it is possible that the otic disc is an example of such a detached island of specialized neuroectodermal material. It can be readily understood, from examination of the sections in figure 11-11, that operative removal of this specialized otic plate would result in complete absence of an otic vesicle on that side of the head, and that the adjacent skin ectoderm, being different in composition, would not have the requisite potentialities to repair the loss. This has become a familiar experiment.

Fig. 11-11. Sections of the otic plate in selected embryos, showing the phases of invagination that characterize this stage. The serial number of the embryo and the number of somites are given in each case. All are enlarged to the same scale.
The specimens selected for illustration in figure 11-11 are especially well preserved and may be accepted as presenting the normal form. In macerated embryos the marked histological difference between skin ectoderm and the neural ectoderm of the otic disc is easily overlooked. In such material the otic plate may be thinned out and its outlines distorted.
In stage 11 it is possible to see cells leaving the otic Page 121 epithelium even more clearly than in stage 10. The basement membrane is disrupted at those sites. Ventral to the otic pit the migrating cells form a characteristic cellular sheath, and they still have the appearance of neural crest cells.
SPECIMENS OF STAGE 11 ALREADY DESCRIBED
13–14 somites, University of Chicago collection, No. H 8. A macerated and damaged specimen. Described by Bartelmez (1922) and also included in the study by Bartelmez and Evans (1926).
13–14 somites, No. 121, Department of Anatomy, Tohoku University, Sendai. Distribution of alkaline phosphatase studied by Mori (1959b).
14 somites, Carnegie No. 12. A pioneer specimen sectioned under poor technical facilities. Described by Mall (1897).
14 somites, Pfannenstiel III, Giessen. Described as embryo No. 6 in the Normentafeln by Keibel and Elze (1908). Representative sections schematically shown in text figure 6, a–w. External form of embryo pictured on plate 5 as figures Vr. and Vv. Embryo described in monograph form by Low (1908). Plastic models are shown by him of external form, central nervous system, digestive system, heart and large vessels. The specimen has two pharyngeal arches, clearly marked. The neural lips are closed rostrally as far as the collicular region.
14 somites, Carnegie No. 4529. A well-preserved specimen, well described by Heuser (1930). More monographs of this standard are needed.
14 somites, embryo von Bulle, Anatomical Collection, Basel. Described by Kollman (1889). Also pictured by Kollman in his Handatlas (1907). Described in the Keibel and Elze Normentafeln as No. 5, and external form pictured on plate 1 as figure IV. Kollman originally reported that there were 13 somites, but his figures show 14 distinct elevations. The closure of the neural tube was probably further rostralward than is shown.
14 somites, Cano embryo, Madrid. Described by Orts-Llorca, Lopez Rodriguez, and Cano Monasterio (1958). Histological condition only fair.
14 somites, Carnegie No. 779. This highly abnormal embryo is included because of the detailed account that has been published of its complete dysraphia (Dekaban, 1963; Dekaban and Bartelmez, 1964).
Approximately 15 somites, collection of First Anatomical Institute of Vienna, embryo Hal. 2. Heart described by Tandler and pharynx by Grosser in Keibel and Mall (1912). Embryo reported as swollen and not suited for cytological studies (Politzer, 1928a).
15 somites, von Spee collection. No 52, Kiel. Camera lucida drawing by Graf Spee before treatment with alcohol,showing right profile of embryo, umbilical vesicle, and connecting stalk. There are 15 discernible somites (Doderlein, 1915, fig. 37). Sections reveal good histological condition of tissues. Used by Evans in Keibel and Mall (1912, fig. 411).
15 somites, University of Chicago collection, No. H 810 (Carnegie No. 8962). Series incomplete, histology fair. Described by Dorland and Bartelmez (1922).
17 somites, Carnegie No. 470. Description, chiefly of central nervous system, published by Bartelmez and Evans (1926).
17 somites, Carnegie No. 4315 (University of Chicago No. H951). Described by Wen (1928) with particular reference to the nervous system. Possibly 18 somites but probably not (Arey, 1938).
17 somites, Carnegie No. 5072. Monographic description, based on three-dimensional reconstructions in addition to the study of the serial sections, published by Atwell (1930).
17 and 19 somites, twins, Toronto collection, No. V and No. VI. Described by Watt (1915).
18 somites, Giglio-Tos, embryo A. Description published by Giglio-Tos (1902). Embryo reported to have 15 segments, but from other evidence it is probably more advanced. The author notes that the caudal neuropore is open, and the reader must infer that the rostral neuropore is closed. The otic invagination appears to be well advanced, although still open. There should therefore be 18 or more somites. The cytological description of the neural tube indicates good preservation. There are no drawings to portray the form of the embryo and the status of organogenesis.
18 somites, Embryological Institute, Vienna, embryo B. Described in part by Sternberg (1927) in a study of the closure of the rostral neuropore. A monographic description of this embryo was published by Politzer (1928a), which establishes it as one of the best representatives of this stage. Unfortunately destroyed.
18 somites, von Spee, Kiel. Only drawings of the external form, right profile and dorsal views, and a sketch showing perforation of the oropharyngeal membrane, are available. The rostral neuropore appears closed. These drawings seem to have been made by Graf Spee and constitute figures 38, 39, and 42 in Döderlein's Handbuch (1915). Apparently the embryo has never been sectioned or studied in detail.
20 somites, Embryological Laboratory of Geneva, embryo Eternod-Delaf. Described in monographic form by Bujard (1913-1914). A well-preserved normal embryo. Description given of its external form. Carefully made plastic and graphic reconstructions reveal the form of the central nervous system, digestive system, coelom, vascular system, and somites. The rostral neuropore is almost closed; there are two pharyngeal arches. It is probable, therefore, that the somitic count is correct.
20 somites, Carnegie No. 2053 A notable description of this embryo was published by Davis (1923). Because of its histological excellence and the care with which it has been studied, this specimen deserves a position of distinction.
Copyright © 1987 Carnegie Institution of Washington. Reproduced on ehd.org with permission.
